| Table of Contents |  |
|
Short Report
| ||||||
| Binding of FAK and HIF-1 during mechanoperception | ||||||
| Patricia Sanz-Ramos1, Iñigo Izal-Azcárate1 | ||||||
|
1Laboratory for Orthopedic Research, University of Navarra, School of Medicine, Pamplona, Spain.
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Sanz-Ramos P, Izal-Azcárate I. Binding of FAK and HIF-1 during mechanoperception. Edorium J Cell Biol 2014;1:12–15. |
|
Abstract
|
|
Aims:
We have suggested the interaction of HIF-1a and FAK, as a mechanism for the influence of low oxygen concentrations in the mechanoperception in chondrocytes. We aimed here, to analyze the binding capacity for HIF-1a and FAK in protein extracts from rat chondrocytes in hypoxia.
Methods: We have used protein extracts from chondrocytes subjected to changes in substrate stiffness for 30 minutes used them for immunoprecipitation and pull down assays. Results: We show the binding capacity for FAK and HIF-1a and demonstrate that it is independent on the phosphorylation of FAK. Conclusion: When in hypoxia, this interaction occurs in chondrocytes that suffer a change in the stiffness and plays a role in the mechanisms triggered by the mechanosensing. | |
|
Keywords:
Chondrocyte, Hypoxia, Mechanosensing, FAK, HIF-1α
| |
|
Introduction
|
|
Aiming to give significant clues concerning the overall mechanism that allows a chondrocyte to behave differently when cultured on soft matrices, we have investigated several process during the mechanoperception cascade occurring in a chondrocyte. In a previous study, we described the participation of integrins (α1, β1 and β3), other focal adhesion (FA) members (Paxillin, RhoA or Src) and a special role for FAK and HIF-1 during hypoxia mechanosensing, suggesting the binding of these two proteins during the process [1]. FAK seems to be the recruiter of the signaling and structural molecules that assemble and form the focal adhesion complexes, functioning as a scaffold for the proteins that integrate them [2] [3]. HIF-1 on its own, has been demonstrated as a key factor in this response to low oxygen concentrations [4] [5] , but nothing has been described concerning a possible implication in the mechanoperception of chondrocytes in hypoxia. We hypothesize that signaling cascades triggered by FAK and HIF-1α culminate in the modulation of cell behavior and in the losing or the maintenance of the final chondrocytic phenotype. We hypothesize here that FAK and HIF-1α, in hypoxic conditions, are able to interact directly and have the capacity to bind. We have proved this binding capacity and analyzed for its dependence on FAK phosphorylation. |
|
Materials and Methods
|
|
Immunoprecipitation and Pull down assay Western Blot analysis |
|
Results |
|
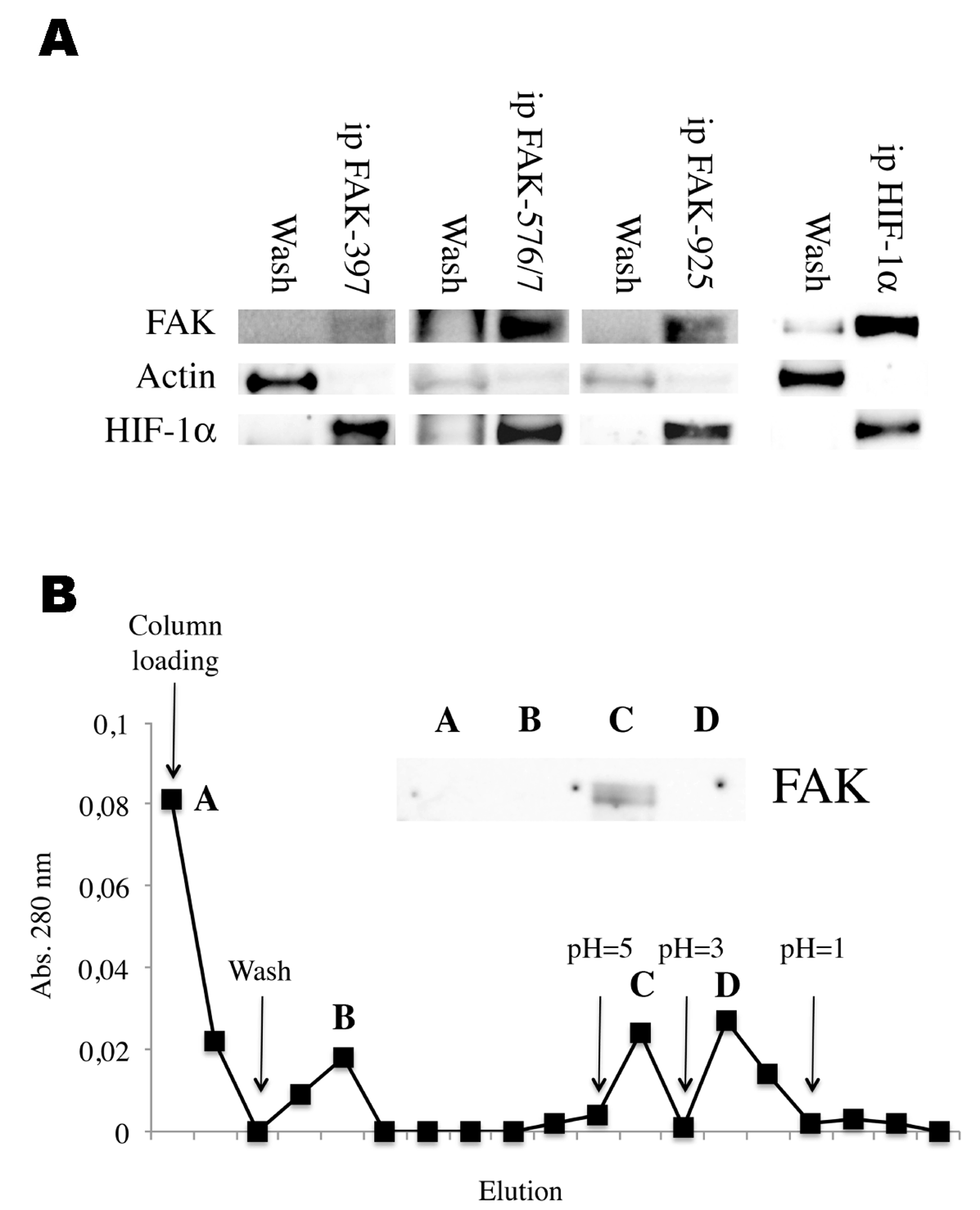
Immunoprecipitation using FAK antibodies were used to determine the binding capacities of HIF and FAK proteins. Hypoxic chondrocytes were placed in collagen hydrogels for 30 minutes and then collected to obtain the total protein content. When HIF-1 antibody was immobilized on magnetic spheres, we detected the coprecipitation of FAK by using western blot analysis of fractions shown in (Figure 1A). We also performed immunoprecipitation using antibodies against different phosphorylated forms of FAK (on tyrosines 397, 576/7 and 925). In all cases, HIF-1 appeared on the precipitated fractions, suggesting the binding capacity independent on the phosphorylated state of FAK (Figure 1A). To confirm this binding, a pull down assay was performed immobilizing HIF-1 on a sepharose column by means of an antibody. The results in Figure 1B show the peaks of proteins measured by absorbance at 280 nm. Peaks selected in the figure were subjected to western blot analysis. We detected the presence of a band corresponding to FAK only after the elution using decreasing pH buffers. According to the figure, we assume peak B for non-specific binding of proteins to the column and D for other proteins that bind to HIF-1α with higher ionic strength than FAK and that have no role in our research. |
|
|
|
Discussion
|
|
Mechanisms that underlie the response to change in the substrate mechanics are not fully described and many data remain to be discovered in order to completely understand what makes a cell behave in the way it does after changes in the stiffness have been applied. This knowledge would give us the possibility to redirect their phenotype as desired. In a previous work, we described the participation of both FAK and HIF-1α in the mechanisms triggered by changes in the matrix rigidity and suggested the possibility of the interaction between these two molecules. Herein, we present data that supports the binding of FAK and HIF-1a during the process of mechanosensing. Results in Figure 1A-B prove the interaction of both molecules in protein extracts from cells 30 minutes after placing them in collagen hydrogels. Moreover, our binding experiments showed the capacity of interaction of both proteins independently of the phosphorylated residues exhibited by FAK. We tested antibodies against FAK-397-P, FAK-576/7-P and FAK-925-P and all of them were capable of precipitating HIF-1α The phosphorylation of FAK is produced in a cascade beginning with the residue 397, which leads to the binding of Src and subsequently, to the phosphorylation in 576/7 and to the interaction with other signaling molecules [6] [7] [8] [9]. A later phosphorylation in residue 925 releases FAK from the FA complex [10]. Our results identify then HIF-1α, as one of those signaling molecules that interact with the FA complex in hypoxia. In a previous work, we showed that FAK was upregulated during mechanoperception in normoxia, but downregulated while in hypoxia, suggesting its implication in the mechanoperception regulation by oxygen content. We also showed that HIF-1α is the responsible for changes in the response to mechanics with oxygen [11]. Despite the biological significance of this interaction remains to be fully described, putting it all together, it seems to take part in the regulation of the response to the mechanics in a hypoxic environment. |
|
Conclusion
|
|
FAK and HIF-1α bind when chondrocytes in a hypoxic environment are subjeceted to change in the substrate stiffness. This interaction does not depend on the phosphorylation of FAK residues Y397, Y576/7 and Y925. |
|
Acknowledgements
|
|
P. Sanz-Ramos received a Fellowship from Asociación de Amigos de la Universidad de Navarra. We would like to thank Juan José Martínez-Irujo for his support in the pull down assay. |
|
References
|
|
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Patricia Sanz-Ramos – Substantial contribution to conception and design of the experiment, Data acquisition, Analysis and interpretation, Drafting the article, Final approval of the version to be published Iñigo Izal-Azcárate – Substantial contribution to conception and design of the experiment, Data acquisition, Analysis and interpretation, Drafting the article, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2014 Patricia Sanz-Ramos et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|